PROFESSIONAL INFORMATION
What is Fitodenta
Fitodenta Dental Surgery Oral Care Kit consists of two products: an oral spray and a topical gel, designed to help alleviate inflammation and swelling following dental surgeries. A clinical survey has demonstrated that when used in combination, these products can result in up to a 40% faster recovery after dental surgeries. It is recommended to use these products as adjunctive oral care alongside traditional treatment methods. It’s important to note that Fitodenta is classified as a cosmetic product and should not be considered medicinal or used for treatment purposes.
Fitodenta Dental Surgery Oral Care Kit consists of:
FITODENTA OROMUCOSAL RELIEF SPRAY for intra-oral use. It is a 20 ml oil spray preparation containing 500 mg of cannabidiol (5%), beta-caryophyllene, plant extracts (cloves, calendula, peppermint, licorice). The other ingredient is medium chain triglyceride oil of vegetable origin.
FITODENTA DEEP RELIEF GEL for topical use. It is a 20 g oil gel containing 400 mg of cannabidiol (2%), plant extracts (cloves, rosemary, peppermint). The other ingredients are medium chain triglyceride oil of vegetable origin, silica.
The products are patent pending, EUIPO Application Number 22212301.0 „Compositions for use in post-operative pain and inflammation treatment“.

When to use
Fitodenta Dental Surgery Oral Care Kit is intended for immediate use following oral or dental surgeries. These products are designed for home use by the patient themselves. The oral spray should be applied to the surgical site on the mucosa and gums, but only when a blood clot has formed to cover the wound. The topical gel should be applied to the face in the area of inflammation and swelling. Both products should be applied simultaneously and can be used for up to 7 days or until complete relief of post-surgery symptoms is achieved.
How to use
FITODENTA OROMUCOSAL RELIEF SPRAY:
2-3 sprays 3 times a day on the irritated gums and area of surgery. The recommended interval is 6 hours.
FITODENTA DEEP RELIEF GEL:
Apply a small amount of gel (1-2 g) topically on the area of surgery and other painful face area:
– on the skin,
– area of swelling,
Apply the gel 3 times a day, on average every 6 hours. Avoid contact with eyes. Wash hands thoroughly after use.
The recommended total duration of use is up to 7 days.
We recommend monitoring patient condition. If you have any questions, please contact us at info@fitodenta.com. Not recommended for children under 18 years old and pregnant women.
FITODENTA DOUBLE-SIDED EFFECT

Source: https://www.physio-pedia.com/Trigeminal_Nerve
The trigeminal nerve is the large sensory nerve that supplies feeling to your face, mouth, eyes, nose and scalp. Nerve impact can result from dental treatments such as dental injections, root canals treatments, insertion of dental implants, removal of teeth, and other surgical treatments. Pain associated with oral and dental surgeries can involve the trigeminal nerve in several ways, as the trigeminal nerve plays a crucial role in transmitting sensory information from the face, mouth, and oral cavity to the brain (1).
- Sensory Innervation: The trigeminal nerve (cranial nerve V) is responsible for providing sensory innervation to various structures in the head and neck, including the oral and dental regions. This nerve carries sensory information related to touch, pain, and temperature from these areas to the brain.
- Nociception: Nociceptors are specialized sensory receptors that detect noxious stimuli or potential tissue damage. These nociceptors are abundant in the oral and dental tissues. When oral or dental surgery is performed, these nociceptors can be stimulated, leading to the perception of pain.
- Surgical Trauma: During oral and dental surgeries, there may be manipulation, cutting, or trauma to the tissues innervated by the trigeminal nerve. This can activate nociceptors and trigger pain signals that are transmitted through the trigeminal nerve to the brain.
- Inflammatory Response: Following surgery, an inflammatory response in the surgical area can contribute to pain. The trigeminal nerve can transmit signals related to the inflammation and tissue damage, causing pain sensations.
- Neuropathic Pain: In some cases, damage to the trigeminal nerve itself during surgery can lead to neuropathic pain. Neuropathic pain is characterized by abnormal, shooting, or burning sensations and may persist even after the surgical site has healed.
- Referred Pain: The trigeminal nerve is also interconnected with other nerves and can contribute to referred pain. Referred pain is when pain is perceived in a different location from the actual source of stimulation. For example, dental pain might be referred to the ear or head due to the complex sensory connections involving the trigeminal nerve.
It’s essential for dental professionals to consider the potential involvement of the trigeminal nerve in the pain process and take appropriate measures to minimize and manage pain effectively for their patients.
- Schiavone M., Ziccardi V.B.: Trigeminal nerve injuries in oral and maxillofacial surgery: a literature review. FOMM, 2021.
Fitodenta Double-Sided Effect:
FITODENTA OROMUCOSAL RELIEF SPRAY (mouth spray) and FITODENTA DEEP RELIEF GEL (topical gel) work synergistically for maximum effectiveness. Apply the oral spray directly to irritated gums and mucosa inside the mouth and the gel topically to the swollen cheek simultaneously. This dual approach targets both cranial/peripheral nerves and swollen tissues, providing efficient inflammation and pain alleviation. The oral spray also alleviates discomfort from dysesthesia following post-surgery peripheral nerve damage. The topical gel also eases symptoms of post-surgery tissue damage, including swelling and hemorrhage. In clinical studies, this combination demonstrated the best results, accelerating post-surgery recovery times by up to 40%.

FITODENTA ANTIMICROBIAL EFFECT
A study was conducted to determine the Minimal Inhibitory Concentration (MIC)* of the Test Item FITODENTA OROMUCOSAL RELIEF SPRAY against Staphylococcus aureus ATCC6538. The study involved a test combining a disk diffusion pattern with the determination of MIC, conducted in three independent replicates, each performed twice.
Based on the results obtained and interpreted according to CLSI M45 guidelines, it can be concluded that the Test Item FITODENTA OROMUCOSAL RELIEF SPRAY exhibits antimicrobial activity with a Minimal Inhibitory Concentration (MIC) observed up to a dilution factor of 1:8 against Staphylococcus aureus, under the experimental conditions employed.
*- Minimal Inhibitory Concentration (MIC) is defined as the highest dilution (or the lowest concentration) of an antimicrobial agent that inhibits the growth of a test microorganism after 24 hours of incubation under standardized conditions.
Safety information, allergen information
The safety assessments of Fitodenta Oromucosal Spray and Fitodenta Topical Gel have been conducted in accordance with industry regulations and guidelines. The products have been found to be safe for consumer use when used as directed.
Safety report FITODENTA OROMUCOSAL SPRAY
Safety report FITODENTA DEEP RELIEF GEL
Certificate of Analysis FITODENTA OROMUCOSAL SPRAY
Certificate of Analysis FITODENTA DEEP RELIEF GEL
Microbial Test Results FITODENTA OROMUCOSAL SPRAY
Microbial Test Results FITODENTA DEEP RELIEF GEL
Safety Data Sheet FITODENTA OROMUCOSAL SPRAY
Safety Data Sheet FITODENTA DEEP RELIEF GEL
Allergen information: Contains plant extracts – cloves, rosemary, calendula, licorice, peppermint.
Suitable for vegans and vegetarians: contains only plant-based ingredients.
Clinical survey information
Compatibility with NSAIDs, Antibiotics and antibacterial agents
Fitodenta products are not only recommended but can also be used alongside NSAIDs and antibiotics as adjunctive oral care. There have been no observed negative interactions between Fitodenta products, NSAIDs, and antibiotics. A clinical study demonstrated that patients who used Fitodenta products in combination with NSAIDs and antibiotics experienced up to a 40% faster recovery after dental surgeries.
Additionally, Fitodenta products can be used in conjunction with antibacterial agents such as chlorhexidine or iodine. To achieve the best results, it is advisable to first apply the antibacterial agent to the affected mouth area and, after a few minutes, apply FITODENTA OROMUCOSAL SPRAY to the same area.
Manufacturing standard and regulatory information
Fitodenta products are manufactured in a compounding pharmacy that has implemented good manufacturing practices. These practices align with the principles of European Parliament and Council of Europe Regulation No. 1223/2009, as well as the legal requirements of the Republic of Lithuania, specifically hygiene norms HN 62:2003 and HN 64-1:2001. This facility is under the regulation and accreditation of the national authorities of the Republic of Lithuania. Fitodenta is classified as a cosmetic product and has been duly notified to the EU Cosmetic Product Notification Portal.
CBD IN DENTISTRY
David, et. al. 2022. Review. Dent. J. 10, 193
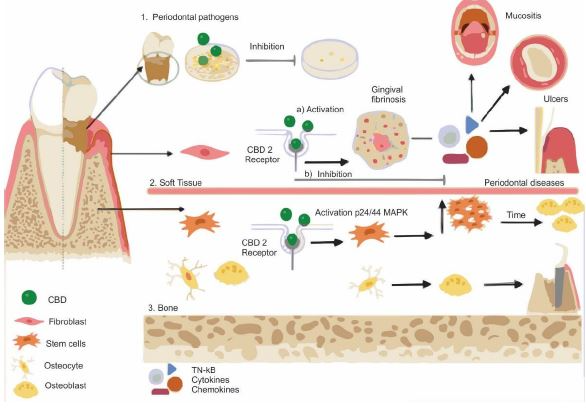
According to literature reports, CBD has several properties which are very important for therapeutic applications in dentistry. Potential therapeutic targets of CBD include gingival ulcers, mucositis, and other adjuncts in periodontal diseases (1). Antiseptic or antimicrobial properties of CBD were shown to be potential for reduction of colonies of periodontal pathogens (2). Stimulation of tissue regeneration potency by CBD is related to CBD-mediated activation of gingival fibrosis which resulted in to elevated production of gingival fibroblast. The effect of elevated levels of gingival fibrosis is explained by activation of CB2 receptor in fibroblast by CBD which results in stimulation of growth factors and decrease of matrix-related metalloproteinases (3). Bone tissue regeneration can be also promoted by CBD. CBD – stimulated mesenchymal cells at the lesion site are more efficiently differentiating into osteoblasts. Here, the action of CBD is related to the activation of p42/44 mitogen-activated protein kinases (MAPK) in mesenchymal cells (4).
Finally, Anti-inflammatory, antioxidant, and analgesic features of CBD are related to an ability of CBD to inhibit or modulate the production of cytokines, chemokines, and pro-inflammatory growth factors. CBD is also interfering with the migration of macrophages and neutrophils (5,6).
- David, C.; Elizalde-Hernández, A.; Barboza, A.S.; Cardoso, G.C.; Santos, M.B.F.; Moraes, R.R. Cannabidiol in Dentistry: A Scoping Review. J.2022, 10, 193.
- Stahl, V.; Vasudevan, K. Comparison of efficacy of cannabinoids versus commercial oral care products in reducing bacterial content from dental plaque: A preliminary observation. Cureus 2020, 12.
- Rawal, S.; Dabbous, M.K.; Tipton, D. Effect of cannabidiol on human gingival fibroblast extracellular matrix metabolism: MMP production and activity, and production of fibronectin and transforming growth factor _. J. Periodontal Res. 2012, 47, 320–329
- Kamali, A.; Oryan, A.; Hosseini, S.; Ghanian, M.H.; Alizadeh, M.; Eslaminejad, M.B.; Baharvand, H. Cannabidiol-loaded microspheres incorporated into osteoconductive scaffold enhance mesenchymal stem cell recruitment and regeneration of critical-sized bone defects. Mater. Sci. Eng. C 2019, 101, 64–75.
- Pisanti, S.; Malfitano, A.M.; Ciaglia, E.; Lamberti, A.; Ranieri, R.; Cuomo, G.; Abate, M.; Faggiana, G.; Proto, M.C.; Fiore, D.; et al.Cannabidiol: State of the art and new challenges for therapeutic applications. Pharmacol. Ther. 2017, 175, 133–150.
- Ossola, C.A.; Surkin, P.N.; Mohn, C.E.; Elverdin, J.C.; Fernández-Solari, J. Anti-inflammatory and osteoprotective effects of cannabinoid-2 receptor agonist HU-308 in a rat model of lipopolysaccharide-induced periodontitis. J. Periodontol. 2016, 87, 725–734.